13-METHYLTETRADECANOIC ACID CAS#: 2485-71-4; ChemWhat Code: 46125
Identification
| Product Name | 13-METHYLTETRADECANOIC ACID |
| IUPAC Name | 13-methyltetradecanoic acid |
| Molecular Structure | 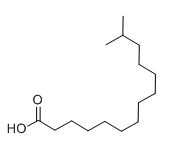 |
| CAS Registry Number | 2485-71-4 |
| EINECS Number | 256-870-9 |
| MDL Number | MFCD00083431 |
| Beilstein Registry Number | No data available |
| Synonyms | 13-methyltetradecanoic acid13-methylmyristic acid13‑methyltetradecanoic acidIsopentadecylic acid13-MTD |
| Molecular Formula | C15H30O2 |
| Molecular Weight | 242.403 |
| InChI | CC(C)CCCCCCCCCCCC(=O)O |
| InChI Key | ZOCYQVNGROEVLU-UHFFFAOYSA-N |
| Canonical SMILES | CC(C)CCCCCCCCCCCC(=O)O |
| Patent Information | ||
| Patent ID | Title | Publication Date |
| CN119390737 | Preparation method of tunicamycin-V and analogues thereof | 2025 |
| US7109364 | Group of anti-cancer compounds with specific structure and their production method | 2006 |
Physical Data
| Appearance | White powder |
| Solubility | No data available |
| Flash Point | No data available |
| Refractive index | No data available |
| Sensitivity | No data available |
| Melting Point, °C | Solvent (Melting Point) |
| 47 – 49 | |
| 51 – 51.9 | acetone |
| 49 – 50 | |
| 43 – 46 |
| Refractive Index | Wavelength (Refractive Index), nm | Temperature (Refractive Index), °C |
| 1.4312 | 589 | 59 |
| Description (Association (MCS)) | Solvent (Association (MCS)) | Partner (Association (MCS)) |
| Formation constant of a complex | aq. phosphate buffer | cytochrome P450BM3 |
| Chromatographic data |
| GC (Gas chromatography) |
| HPLC (High performance liquid chromatography) |
| TLC (Thin layer chromatography) |
| Description (Crystal Phase) | Comment (Crystal Phase) |
| Interplanar spacing | 29.8 Angstroem. |
Spectra
| Description (NMR Spectroscopy) | Nucleus (NMR Spectroscopy) | Solvents (NMR Spectroscopy) | Temperature (NMR Spectroscopy), °C |
| Chemical shifts, Spectrum | 1H | chloroform-d1 | |
| Chemical shifts, Spectrum | 1H | chloroform-d1 | 20 |
| Chemical shifts | 1H | CD3OD |
| Description (IR Spectroscopy) | Solvent (IR Spectroscopy) | Original Text (IR Spectroscopy) | Comment (IR Spectroscopy) |
| Bands | IR (film) = 2917, 2850, 1698, 1472 cm-1 | film | |
| ATR (attenuated total reflectance), Intensity of IR bands, Bands | |||
| Bands | neat (no solvent, solid phase) | ||
| Bands | chloroform |
| Solvent (UV/VIS Spectroscopy) | Absorption Maxima (UV/VIS), nm | Log epsilon |
| dichloromethane | 222, 224, 230 | 2.05, 2.25, 2.25 |
Route of Synthesis (ROS)
| Conditions | Yield |
| With sodium hydroxide In ethanol at 40℃; for 4h; | 89% |
| With potassium hydroxide In ethanol | |
| With sodium hydroxide In ethanol at 20℃; | 99.7% |
Safety and Hazards
| Pictogram(s) |  |
| Signal | Warning |
| GHS Hazard Statements | H315 (100%): Causes skin irritation [Warning Skin corrosion/irritation] H319 (100%): Causes serious eye irritation [Warning Serious eye damage/eye irritation] H335 (100%): May cause respiratory irritation [Warning Specific target organ toxicity, single exposure; Respiratory tract irritation] Information may vary between notifications depending on impurities, additives, and other factors. |
| Precautionary Statement Codes | P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, and P501 (The corresponding statement to each P-code can be found at the GHS Classification page.) |
Other Data
| Transportation | NONH for all modes of transport |
| Under the room temperature and away from light | |
| HS Code | No data available |
| Storage | Under the room temperature and away from light |
| Shelf Life | 2 years |
| Market Price | USD |
| Druglikeness | |
| Lipinski rules component | |
| Molecular Weight | 242.402 |
| logP | 6.94 |
| HBA | 2 |
| HBD | 1 |
| Matching Lipinski Rules | 3 |
| Veber rules component | |
| Polar Surface Area (PSA) | 37.3 |
| Rotatable Bond (RotB) | 12 |
| Matching Veber Rules | 1 |
| Use Pattern |
| Antitumor/Bioactivity Research (Core Application): This substance exhibits significant anticancer activity and can induce tumor cell apoptosis. Its effectiveness has been verified in various cancer cells (such as bladder cancer and lymphoma). |
| Biochemistry/Metabolic Research: As a branched-chain fatty acid (BCFA) model molecule, it is used to study: lipid metabolism and microbial metabolic pathways. |
| Microbial and Biomaterials Research: It can be used as a substrate for: the synthesis of polyhydroxyalkanoates (PHA) biomaterials. |
| It is used to study: biodegradable plastics and the structural regulation of biopolymers. |
| Modulation of growth rate between L4 and adult stage |
Buy Reagent | |
| No reagent supplier? | Send quick inquiry to ChemWhat |
| Want to be listed here as a reagent supplier? (Paid service) | Click here to contact ChemWhat |
Approved Manufacturers | |
| Caming Pharmaceutical Ltd | http://www.caming.com/ |
| Want to be listed as an approved manufacturer (Requires approvement)? | Please download and fill out this form and send back to approved-manufacturers@chemwhat.com |
Other Suppliers | |
| Watson International Limited | Visit Watson Official Website |
Contact Us for Other Help | |
| Contact us for other information or services | Click here to contact ChemWhat |