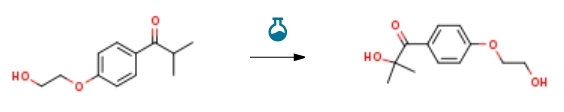
2-Hydroxy-4′-(2-hydroxyethoxy)-2-methylpropiophenone CAS#: 106797-53-9; ChemWhat Code: 204919
Identification
| Product Name | 2-Hydroxy-4′-(2-hydroxyethoxy)-2-methylpropiophenone |
| IUPAC Name | 2-hydroxy-1-[4-(2-hydroxyethoxy)phenyl]-2-methylpropan-1-one |
| Molecular Structure | |
| CAS Registry Number | 106797-53-9 |
| MDL Number | MFCD00085267 |
| Synonyms | 106797-53-9 2-Hydroxy-4′-(2-hydroxyethoxy)-2-methylpropiophenone 2-Hydroxy-1-(4-(2-hydroxyethoxy)phenyl)-2-methylpropan-1-one Darocur 2959 2-hydroxy-1-[4-(2-hydroxyethoxy)phenyl]-2-methylpropan-1-one Irgacure 2959 2-Hydroxy-1-[4-(2-hydroxyethoxy)phenyl]-2-methyl-1-propanone 1-Propanone, 2-hydroxy-1-[4-(2-hydroxyethoxy)phenyl]-2-methyl- MFCD00085267 DTXSID6044777 I2959 2-hydroxy-4-(2-hydroxyethoxy)-2-methylpropiophenone 2-Hydroxy-1-(4-(2-hydroxyethoxy)phenyl)-2-methyl-1-propanone 1-Propanone, 2-hydroxy-1-(4-(2-hydroxyethoxy)phenyl)-2-methyl- 2-Hydroxy-4 inverted exclamation marka-(2-hydroxyethoxy)-2-methylpropiophenone 4-(2-Hydroxyethoxy)phenyl 2-hydroxy-2-propyl ketone 1-[4-(2-hydroxyethoxy)phenyl]-2-hydroxy-2-methyl-1-propanone photoinitiator 2959 2-hydroxy-4′-(2-hydroxyethoxy)-2-methyl propiophenone Darocur 2595 IRGACURE2959 SCHEMBL36443 CHEMBL3188617 DTXCID4024777 IRGC 2959 CHEBI:170047 2-hydroxy-1-[4-(2-hydroxyethoxy)phenyl]-2-methyl-propan-1-one 2-hydroxy-2-methyl-1-[4-(2-hydroxyethoxy)phenyl]propan-1-one IRG 2959 ZLI 2959 Tox21_301074 AKOS015889779 CS-W014224 DC 2959 HY-W013508 UV 2959 NCGC00248279-01 NCGC00254975-01 DS-17470 SY052526 DB-059511 CAS-106797-53-9 H1361 NS00014925 D 2959 D70491 I 2959 J-001651 2-Hydroxy-4\’-(2-hydroxyethoxy)-2-methylpropiophenone 4-(2-hydroxyethoxy)phenyl (2-hydroxy-2-propyl) ketone 1-(4-(2-hydroxyethoxy)phenyl)-2-hydroxy-2-methylpropan-1-one 2-hydroxy-1-[4-(hydroxyethoxy)phenyl]-2-methyl-1-propanone 2-Hydroxy-4′-(2-hydroxyethoxy)-2-methylpropiophenone, 98% 1-[4-(2-Hydroxyethoxy)phenyl]-2-hydroxy-2-methyl-1-propan-1-one 2-hydroxy-1-[4-(2-hydroxy-ethoxy)phenyl]-2-methylpropan-1-one 2-Hydroxy-4 inverted exclamation mark -(2-hydroxyethoxy)-2-methylpropiophenone |
| Molecular Formula | C12H16O4 |
| Molecular Weight | 224.25 |
| InChI | InChI=1S/C12H16O4/c1-12(2,15)11(14)9-3-5-10(6-4-9)16-8-7-13/h3-6,13,15H,7-8H2,1-2H3 |
| InChI Key | GJKGAPPUXSSCFI-UHFFFAOYSA-N |
| Isomeric SMILES | CC(C)(C(=O)C1=CC=C(C=C1)OCCO)O |
| Patent Information | ||
| Patent ID | Title | Publication Date |
| CN109896942 | Preparation method of alpha-hydroxyketone photoinitiator | 2019 |
| CN110563588, | Mono-cinnamate or dicinnamate photoinitiators as well as preparation method and application thereof | 2019 |
| CN107739303 | Alpha-hydroxy ketone photoinitiator preparation method | 2018 |
| CN107868034 | Photoinitiators applicable to UV-LED photocuring | 2018 |
| CN104710256 | Α – hydroxy ketone compound low priced high-efficient synthetic method | 2017 |
| CN103288873 | Sulfonyl or quinonyl functionalized acyl phosphine oxidation compound | 2017 |
Physical Data
| Melting Point, °C |
| 88 – 90 |
Spectra
| Description (NMR Spectroscopy) | Nucleus (NMR Spectroscopy) | Solvents (NMR Spectroscopy) | Frequency (NMR Spectroscopy), MHz |
| Chemical shifts | 1H | chloroform-d1 | 400 |
| Chemical shifts, Spectrum | 1H | ||
| Chemical shifts, Spectrum | 1H | chloroform-d1 | 400 |
| Chemical shifts, Spectrum | 13C | chloroform-d1 | 100 |
| Description (IR Spectroscopy) | Solvent (IR Spectroscopy) |
| Spectrum | potassium bromide |
| Bands, Spectrum | |
| Bands | neat liquid |
| Description (UV/VIS Spectroscopy) | Solvent (UV/VIS Spectroscopy) | Absorption Maxima (UV/VIS), nm |
| Spectrum | water, methanol | |
| Spectrum | dimethyl sulfoxide | 280 |
| Spectrum | acetonitrile | |
| Spectrum | water | |
| Band assignment, Spectrum | methanol |
Route of Synthesis (ROS)
| Conditions | Yield |
| With N-Bromosuccinimide; dimethyl sulfoxide at 100℃; for 24h; | 92% |
| With N-Bromosuccinimide; dimethyl sulfoxide at 60℃; for 24h; Schlenk technique; Experimental Procedure 26 Embodiment 262 – hydroxy -4 ‘ – (2 – hydroxy ethoxy group) -2 – methylpropiophenone preparation Taking a 25 ml Schlenk reaction tube, adding N – bromo succimide (NBS) 18 mg (0.1mmol) as catalyst, 4′ – (2 – hydroxy ethoxy group) -2 – methylpropiophenone 105 mg (0.5mmol), dimethyl sulfoxide (DMSO) 1 ml as the oxidizing agent, carbonylating and solvent, for 100 °C stirring for 24 hours. After the reaction by adding ethyl acetate 15 ml, salt water 3 ml, ethyl acetate 3 times, the combined organic phase, column chromatography separation to obtain 2 – hydroxy -4 ‘ – (2 – hydroxy ethoxy group) -2 – methylpropiophenone pure product 103 mg, yield 92%. | 92% |
| With oxygen; caesium carbonate; triethyl phosphite In dimethyl sulfoxide at 25℃; under 760.051 Torr; for 24h; Schlenk technique; | 75% |
| With dihydrogen peroxide In dichloromethane at 30℃; Microwave irradiation; Experimental Procedure 3 Preparation of 2-hydroxy-1-(4-(2-hydroxyethoxy)phenyl)-2-methyl-1-propanone 44.1 g (0.5 mol) of 4-(2-hydroxyethoxy)isobutyrophenone, 300 mL of dichloroethane and 283.3 g (2.5 mol) of 30% hydrogen peroxide solution were uniformly mixed and placed in a microwave reactor (power of 500 W). , Stir vigorously, and control the reaction temperature at about 30 °C reaction, using TLC or GC to monitor the reaction, the reaction is complete, the microwave radiation, the reaction system was slowly added sodium bisulfite aqueous solution (containing 146g of sodium bisulfite), The reaction was extirpated for 2 h, allowed to stand, separated, and the organic phase was washed with water. The solvent was recovered by decompression at atmospheric pressure, and the residue was recrystallized from methanol.The white solid 2-hydroxy-1-(4-(2-hydroxyethoxy)phenyl)-2-methyl-1-propanone 79.6 g was obtained, HPLC content was 99.1%, yield 71.0%. | 71% |
Safety and Hazards
| GHS Hazard Statements | Not Classified |
Source: European Chemicals Agency (ECHA)
License Note: Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: “Source: European Chemicals Agency, http://echa.europa.eu/”. Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page.
License URL: https://echa.europa.eu/web/guest/legal-notice
Record Name: (1-Cyano-2-ethoxy-2-oxoethylidenaminooxy)dimethylamino-morpholino-carbenium hexafluorophosphate
URL: https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/213446
Description: The information provided here is aggregated from the “Notified classification and labelling” from ECHA’s C&L Inventory. Read more: https://echa.europa.eu/information-on-chemicals/cl-inventory-database
Other Data
| Druglikeness | |
| Lipinski rules component | |
| Molecular Weight | 224.257 |
| logP | 0.704 |
| HBA | 3 |
| HBD | 2 |
| Matching Lipinski Rules | 4 |
| Veber rules component | |
| Polar Surface Area (PSA) | 66.76 |
| Rotatable Bond (RotB) | 5 |
| Matching Veber Rules | 2 |
| Use Pattern |
| 2-Hydroxy-4′-(2-hydroxyethoxy)-2-methylpropiophenone CAS#: 106797-53-9 98%min Often used as UV curable coatings and inks, low odor, and can be used for food packaging and medical packaging. UV Absorption Peak: 274,320 nm. |
Buy Reagent | |
| No reagent supplier? | Send quick inquiry to ChemWhat |
| Want to be listed here as a reagent supplier? (Paid service) | Click here to contact ChemWhat |
Approved Manufacturers | |
| Want to be listed as an approved manufacturer (Requires approvement)? | Please download and fill out this form and send back to approved-manufacturers@chemwhat.com |
Other Suppliers | |
| Watson International Limited | Visit Watson Official Website |
Contact Us for Other Help | |
| Contact us for other information or services | Click here to contact ChemWhat |