2′,3′,5′-Tri-O-acetyluridine CAS#: 4105-38-8; ChemWhat Code: 96969
Identification
| Product Name | 2′,3′,5′-Tri-O-acetyluridine CAS#: 4105-38-8 |
| IUPAC Name | [(2R,3R,4R,5R)-3,4-diacetyloxy-5-(2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methyl acetate |
| Molecular Structure | 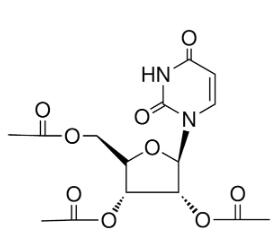 |
| CAS Registry Number | 462-08-8 |
| EINECS Number | 223-881-5 |
| MDL Number | MFCD07798677 |
| Beilstein Registry Number | No data available |
| Synonyms | Tri-O-acetyluridineuridine triacetate2′,3′,5′-tri-O-acetyluridine2′,3′,5′-tri-O-acetyluridine |
| Molecular Formula | C15H18N2O9 |
| Molecular Weight | 370.319 |
| InChI | InChI=1S/C15H18N2O9/c1-7(18)23-6-10-12(24-8(2)19)13(25-9(3)20)14(26-10)17-5-4-11(21)16-15(17)22/h4-5,10,12-14H,6H2,1-3H3,(H,16,21,22)/t10-,12-,13-,14-/m1/s1 |
| InChI Key | AUFUWRKPQLGTGF-FMKGYKFTSA-N |
| Canonical SMILES | CC(=O)OC[C@H]1OC@@HC@H[C@@H]1OC(C)=O |
| Patent Information | ||
| Patent ID | Title | Publication Date |
| WO2022/89612 | USE OF URIDINE DERIVATIVE IN PREPARATION OF MEDICAMENT | 2022 |
| CN105503742 | Synthetic method of difluoroalkyl substituted uracil, uridine or uridylic acid | 2016 |
| US2001/5719 | COMPOSITIONS AND METHODS FOR TREATMENT OF MITOCHONDRIAL DISEASES | 2001 |
Physical Data
| Appearance | White or almost white crystalline powder |
| Solubility | No data available |
| Flash Point | No data available |
| Refractive index | No data available |
| Sensitivity | No data available |
| Melting Point, °C |
| 106 – 108 |
| 133 |
| 127 – 128 |
| 128.3 – 129.7 |
| Description (Association (MCS)) | Solvent (Association (MCS)) | Temperature (Association (MCS)), °C | Partner (Association (MCS)) |
| Association with compound | CDCl3 | -20 – 20 | 5′-acetyl-2′,3′-isopropylideneadenosine |
| NMR spectrum of the complex | CDCl3 | -20 – 20 | 5′-acetyl-2′,3′-isopropylideneadenosine |
| Stability constant of the complex with … | CDCl3 | 24.9 | 2,12-bis(9-anthracenyl)-7-(4-(dimethylamino)phenyl)-5,6,8,9-tetrahydro-1,13-diazadibenzanthracene-14-carboxylic acid |
| Further physical properties of the complex | CDCl3 | 24.9 | 2,12-bis(9-anthracenyl)-7-(4-(dimethylamino)phenyl)-5,6,8,9-tetrahydro-1,13-diazadibenzanthracene-14-carboxylic acid |
| NMR spectrum of the complex | CDCl3 | N,N’-2,6-pyridinediylbis |
| Chromatographic data |
| TLC (Thin layer chromatography) |
Spectra
| Description (NMR Spectroscopy) | Nucleus (NMR Spectroscopy) | Solvents (NMR Spectroscopy) | Temperature (NMR Spectroscopy), °C | Original Text (NMR Spectroscopy) |
| Chemical shifts, Spectrum | 1H | dimethylsulfoxide-d6 | 25 | |
| Chemical shifts, Spectrum | 13C | dimethylsulfoxide-d6 | 25 | |
| Chemical shifts | 1H | chloroform-d1 | 1H NMR (500 MHz, CDCI3) 5 8.69 (s, XH), 7.33 (d, J = 8.2 Hz, 1H), 5.97 (d, J = 4.4 Hz, 1H), 5.73 (d, J = 8.1 Hz, 1H), 5.28 – 5.26 (m, 2H), 4.31 – 4.26 (m, 3H), 2.08 (s, 3H), 2.07 (s, 3H), 2.04 (s, 3H) ppm. | |
| Chemical shifts | 13C | chloroform-d1 | 13C NMR (126 MHz, CDCI3) 5 170.14, 169.66, 162.50, 150.03, 139.29, 103.41, 87.49, 79.96, 72.74, 70.19, 63.13, 20.80, 20.53, 20.44 ppm. | |
| Spectrum | 1H | chloroform-d1 | ||
| Chemical shifts | 1H | chloroform-d1 |
| Description (IR Spectroscopy) | Solvent (IR Spectroscopy) | Comment (IR Spectroscopy) |
| Bands | neat (no solvent) | |
| Bands | film | |
| Bands | CHCl3 | 3385 – 1636 cm**(-1) |
| IR |
| Description (Mass Spectrometry) |
| electrospray ionisation (ESI), spectrum |
| high resolution mass spectrometry (HRMS), MALDI (Matrix assisted laser desorption ionization), IT (ion trap), spectrum |
| high resolution mass spectrometry (HRMS), electrospray ionisation (ESI), time-of-flight mass spectra (TOFMS), liquid chromatography mass spectrometry (LCMS), spectrum |
| high resolution mass spectrometry (HRMS), electrospray ionisation (ESI), spectrum |
| high resolution mass spectrometry (HRMS), electrospray ionisation (ESI), time-of-flight mass spectra (TOFMS), spectrum |
| Description (UV/VIS Spectroscopy) |
| UV/VIS |
Route of Synthesis (ROS)
| Conditions | Yield |
| With ammonium cerium(IV) nitrate; lithium chloride In acetic acid; acetonitrile at 80℃; for 6h; other conditions investigated; | 94% |
| With ammonium cerium (IV) nitrate; lithium chloride In acetonitrile at 80℃; for 44h; | 89% |
| With sodium hydrogencarbonate; lithium chloride In acetonitrile Ambient temperature; electrolysis (Pt electrodes, SCE reference, LiClO4 as supporting electrolyte); | 88% |
| With ammonium cerium (IV) nitrate; lithium chloride In acetic acid; acetonitrile at 80℃; for 8h; | 82% |
| With ammonium cerium (IV) nitrate; 1-ethylene glycol monomethyl ether-3-methylimidazolium methanesulfonate; lithium chloride at 80℃; for 2h; | 32% |
Safety and Hazards
| GHS Hazard Statements | Not Classified |
Other Data
| Transportation | NONH for all modes of transport |
| Under the room temperature and away from light | |
| HS Code | No data available |
| Storage | Store at room temperature, Sealed and away from light. |
| Shelf Life | 3 years |
| Market Price | USD |
| Druglikeness | |
| Lipinski rules component | |
| Molecular Weight | 370.316 |
| logP | -0.755 |
| HBA | 11 |
| HBD | 1 |
| Matching Lipinski Rules | 3 |
| Veber rules component | |
| Polar Surface Area (PSA) | 137.54 |
| Rotatable Bond (RotB) | 8 |
| Matching Veber Rules | 2 |
| Use Pattern |
| Pharmaceuticals |
| preventing cutaneous toxicity which is associated with administration of 5-fluorouracil in a mammalian subject |
| preventing mucositis due to cancer chemotherapy in a mammalian subject in combination with thymidine diacetate |
| treating cutaneous toxicity which is associated with administration of capecitabine in a mammalian subject |
Buy Reagent | |
| No reagent supplier? | Send quick inquiry to ChemWhat |
| Want to be listed here as a reagent supplier? (Paid service) | Click here to contact ChemWhat |
Approved Manufacturers | |
| Caming Pharmaceutical Ltd | http://www.caming.com/ |
| Want to be listed as an approved manufacturer (Requires approvement)? | Please download and fill out this form and send back to approved-manufacturers@chemwhat.com |
Other Suppliers | |
| Watson International Limited | Visit Watson Official Website |
Contact Us for Other Help | |
| Contact us for other information or services | Click here to contact ChemWhat |