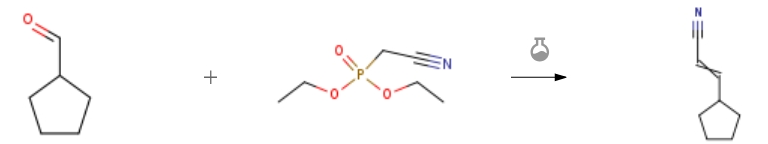
3-CYCLOPENTYLACRYLONITRILE CAS#: 591769-05-0; ChemWhat Code: 900475
Identification
Physical Data
| Appearance | Colorless to pale yellow transparent liquid |
Spectra
| Description (NMR Spectroscopy) | Nucleus (NMR Spectroscopy) | Solvents (NMR Spectroscopy) | Frequency (NMR Spectroscopy), MHz |
| 1H | dimethylsulfoxide-d6 | 300 |
Route of Synthesis (ROS)
| Conditions | Yield |
| With HT-O-t-Bu In N,N-dimethyl-formamide for 2.5h; Wadsworth-Emmons reaction; Heating; | 98% |
| Stage #1: Diethyl cyanomethylphosphonate With potassium tert-butylate In tetrahydrofuran at 0 – 20℃; Stage #2: cyclopentanealdehyde In tetrahydrofuran at 0 – 20℃; for 64h; Experimental Procedure 67.1 Step 1. (2E)- and (2Z)-3-Cyclopentylacrylonitrile Step 1. (2E)- and (2Z)-3-Cyclopentylacrylonitrile To a solution of 1.0 M potassium tert-butoxide in THF (235 mL) at 0 °C was added dropwise a solution of diethyl cyanomethylphosphonate (39.9 mL, 0.246 mol) in THF (300 mL). The cold bath was removed and the reaction was warmed to room temperature followed by recooling to 0 °C, at which time a solution of cyclopentanecarbaldehyde (22.0 g, 0.224 mol) in THF (60 mL) was added dropwise. The bath was removed and the reaction warmed to ambient temperature and stirred for 64 hours. The mixture was partitioned between diethyl ether and water, the aqueous was extracted with three portions of ether, followed by two portions of ethyl acetate. The combined extracts were washed with brine, then dried over sodium sulfate, filtered and concentrated in vacuo to afford a mixture containing 24.4 g of olefin isomers which was used without further purification (89%). 1H NMR (400 MHz, CDCl3): δ 6.69 (dd, 1H, trans olefin), 6.37 (t, 1H, cis olefin), 5.29 (dd, 1H, trans olefin), 5.20 (d, 1H, cis olefin), 3.07-2.95 (m, 1H, cis product), 2.64-2.52 (m, 1H, trans product), 1.98-1.26 (m, 16H). | 89% |
| Stage #1: Diethyl cyanomethylphosphonate With potassium tert-butylate In tetrahydrofuran at 20℃; for 3h; Cooling with ice; Stage #2: cyclopentanealdehyde In tetrahydrofuran at 0 – 20℃; Experimental Procedure 1.3 (3) Preparation of compound (5a) 3-cyclopentaneacrylonitrile Under ice-cooling conditions, 5.9g of diethyl cyanomethylphosphonate was dissolved in 100mL anhydrous tetrahydrofuran. 5.6g of potassium tert-butoxide was added portionwise in the solution and was stirred at room temperature for three hours. The temperature was reduced to 0°C and cyclopentanecarbaldehyde was added dropwise to the reaction solution and reacted overnight at room temperature. TLC was used to monitor reaction completion. 100ml of saturated ammonium chloride solution was added to quench the reaction. After distilling off the solvent, the residue was extracted with ethyl acetate then washed with water. The organic phase was washed with 100mL each of saturated sodium chloride solution 3 times. The organic phase was dried over anhydrous anhydrous magnesium sulfate overnight. The mixture was filtered and the solvent was distilled off under reduced pressure to give a crude product. The product was purified by silica gel column chromatography (petroleum ether:ethyl acetate = 80:1) to give 2.5g of intermediate 5a as a colorless liquid, yield: 67%. | 67% |
Safety and Hazards
| Pictogram(s) |     |
| Signal | Danger |
| GHS Hazard Statements | H227 (66.67%): Combustible liquid [Warning Flammable liquids] H301 (100%): Toxic if swallowed [Danger Acute toxicity, oral] H311 (33.33%): Toxic in contact with skin [Danger Acute toxicity, dermal] H314 (33.33%): Causes severe skin burns and eye damage [Danger Skin corrosion/irritation] H331 (33.33%): Toxic if inhaled [Danger Acute toxicity, inhalation] H351 (33.33%): Suspected of causing cancer [Warning Carcinogenicity] H411 (33.33%): Toxic to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard] H412 (66.67%): Harmful to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard] |
| Precautionary Statement Codes | P203, P210, P260, P261, P262, P264, P270, P271, P273, P280, P301+P316, P301+P330+P331, P302+P352, P302+P361+P354, P304+P340, P305+P354+P338, P316, P318, P321, P330, P361+P364, P363, P370+P378, P391, P403, P403+P233, P405, and P501 (The corresponding statement to each P-code can be found at the GHS Classification page.) |
Other Data
| Druglikeness | |
| Lipinski rules component | |
| Molecular Weight | 121.182 |
| logP | 2.61 |
| HBA | 1 |
| HBD | 0 |
| Matching Lipinski Rules | 4 |
| Veber rules component | |
| Polar Surface Area (PSA) | 23.79 |
| Rotatable Bond (RotB) | 1 |
| Matching Veber Rules | 2 |
| Use Pattern |
| 3-CYCLOPENTYLACRYLONITRILE CAS#: 591769-05-0, as an intermediate for Ruxolitinib, plays a crucial role in the synthesis of Ruxolitinib. Ruxolitinib is a selective Janus kinase (JAK1 and JAK2) inhibitor, primarily used to treat myelofibrosis and polycythemia vera. Ruxolitinib can inhibit the proliferation and survival of tumor cells, thereby slowing the growth of tumors. Additionally, by inhibiting the JAK-STAT pathway, it can reduce inflammation caused by tumors, alleviating symptoms. The cyclopentyl structure in 3-Cyclopentylacrylonitrile is a crucial part of the Ruxolitinib molecule, essential for the drug’s activity and stability. This unique structure helps Ruxolitinib achieve high selectivity and efficacy in targeting JAK1 and JAK2 kinases, enhancing its therapeutic effect. |
Buy Reagent | |
| No reagent supplier? | Send quick inquiry to ChemWhat |
| Want to be listed here as a reagent supplier? (Paid service) | Click here to contact ChemWhat |
Approved Manufacturers | |
| Caming Pharmaceutical Limited | http://www.caming.com/ |
| Want to be listed as an approved manufacturer (Requires approvement)? | Please download and fill out this form and send back to approved-manufacturers@chemwhat.com |
Other Suppliers | |
| Watson International Limited | Visit Watson Official Website |
Contact Us for Other Help | |
| Contact us for other information or services | Click here to contact ChemWhat |