5,10,15,20-TETRAKIS(4-METHOXYPHENYL)-21H,23H-PORPHINE IRON(III) CHLORIDE CAS#: 36995-20-7; ChemWhat Code: 379755
Identification
| Product Name | 5,10,15,20-TETRAKIS(4-METHOXYPHENYL)-21H,23H-PORPHINE IRON(III) CHLORIDE |
| IUPAC Name | iron(3+);methoxybenzene;10,15,20-tris(4-methoxyphenyl)-21,22-dihydroporphyrin;dichloride |
| Molecular Structure |  |
| CAS Registry Number | 36995-20-7 |
| MDL Number | MFCD00012405 |
| Synonyms | 36995-20-7 Iron(3+);methoxybenzene;10,15,20-tris(4-methoxyphenyl)-21,22-dihydroporphyrin;dichloride 5,10,15,20-Tetrakis(4-methoxyphenyl)-21H,23H-porphine iron(III) chloride YSWG546 DB-049051 |
| Molecular Formula | C48H39Cl2FeN4O4 |
| Molecular Weight | 862.6 |
| InChI | InChI=1S/C41H32N4O3.C7H7O.2ClH.Fe/c1-46-30-12-4-25(5-13-30)39-33-18-10-28(42-33)24-29-11-19-34(43-29)40(26-6-14-31(47-2)15-7-26)36-21-23-38(45-36)41(37-22-20-35(39)44-37)27-8-16-32(48-3)17-9-27;1-8-7-5-3-2-4-6-7;;;/h4-24,42-43H,1-3H3;3-6H,1H3;2*1H;/q;-1;;;+3/p-2 |
| InChI Key | ZKOPIALJZKUWLE-UHFFFAOYSA-L |
| Isomeric SMILES | COC1=CC=[C-]C=C1.COC1=CC=C(C=C1)C2=C3C=CC(=CC4=CC=C(N4)C(=C5C=CC(=N5)C(=C6C=CC2=N6)C7=CC=C(C=C7)OC)C8=CC=C(C=C8)OC)N3.[Cl-].[Cl-].[Fe+3] |
| Patent Information | ||
| Patent ID | Title | Publication Date |
| CN108947846 | A preparing method of p-nitroacetophenone | 2018 |
| CN106083867 | Method for preparing modified metalloporphyrin and application thereof in catalytic oxidation desulfurization | 2016 |
Physical Data
| Appearance | Dark blue or purple powder |
| Melting Point, °C | Solvent (Melting Point) |
| 300 | dichloromethane, methanol |
| Description (Association (MCS)) | Partner (Association (MCS)) |
| Adsorption | carbon |
| Adsorption to title compound | oxygen |
Spectra
| Description (NMR Spectroscopy) | Nucleus (NMR Spectroscopy) | Solvents (NMR Spectroscopy) | Temperature (NMR Spectroscopy), °C | Frequency (NMR Spectroscopy), MHz |
| Chemical shifts, Spectrum | 1H | chloroform-d1 | 400.1 | |
| Spectrum | 1H | not given | 25 | |
| 1H | chloroform-d1 | 26 |
| Description (IR Spectroscopy) | Solvent (IR Spectroscopy) |
| Bands, Spectrum | potassium bromide |
| Bands | potassium bromide |
| Bands | |
| Bands | KBr |
| Spectrum | nujol |
| Bands | nujol |
| Description (UV/VIS Spectroscopy) | Solvent (UV/VIS Spectroscopy) | Absorption Maxima (UV/VIS), nm |
| Spectrum | dichloromethane | 385, 422, 513, 578, 700 |
| Band assignment | dichloromethane | 421, 511, 572, 698 |
| Spectrum | tetrahydrofuran | 414, 578, 620 |
| Spectrum | supporting information | 425, 502, 581, 695 |
| 423.5 | ||
| 420, 509, 572 |
Route of Synthesis (ROS)
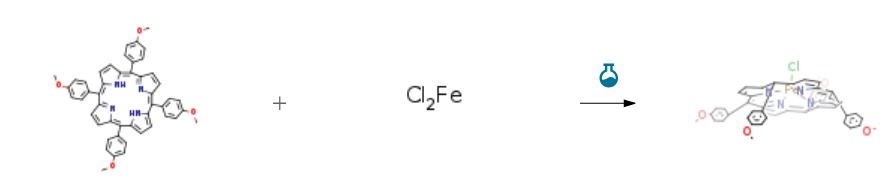
| Conditions | Yield |
| Stage #1: 5,10,15,20-tetrakis(4-methoxyphenyl)porphyrin; iron(II) chloride In tetrahydrofuran for 20h; Reflux; Inert atmosphere; Darkness; Stage #2: With air In tetrahydrofuran at 20℃; for 0.333333h; Darkness; Experimental Procedure General procedure: The corresponding free-base porphyrin 2 and FeCl2 were dissolved in dry THF and the reaction mixture thus obtained was refluxed under argon and under protection from light for 20 h. After cooling of the solution to room temperature, the mixture was exposed to open air under further stirring for 20 min. The solvent was evaporated and the obtained residue was purified by flash chromatography on silica gel, using CH2Cl2/MeOH as mobile phase, in order to remove unreacted FeCl2. Observation of decay of Soret peak in measured UV-vis spectra permitted to monitor completion of iron insertion.20a | 90% |
Safety and Hazards
| GHS Hazard Statements | Not Classified |
Other Data
| Shelf Life | 1 year |
| Market Price |
| Druglikeness | |
| Lipinski rules component | |
| Molecular Weight | 824.138 |
| logP | 15.284 |
| HBA | 4 |
| HBD | 0 |
| Matching Lipinski Rules | 2 |
| Veber rules component | |
| Polar Surface Area (PSA) | 56.64 |
| Rotatable Bond (RotB) | 8 |
| Matching Veber Rules | 2 |
| Use Pattern |
| 5,10,15,20-TETRAKIS(4-METHOXYPHENYL)-21H,23H-PORPHINE IRON(III) CHLORIDE is a metalloporphyrin compound with unique optical, electrochemical, and catalytic properties. It is primarily used in the following areas.This metalloporphyrin compound is commonly used as a catalyst in oxidation reactions. |
Buy Reagent | |
| No reagent supplier? | Send quick inquiry to ChemWhat |
| Want to be listed here as a reagent supplier? (Paid service) | Click here to contact ChemWhat |
Approved Manufacturers | |
| Want to be listed as an approved manufacturer (Requires approvement)? | Please download and fill out this form and send back to approved-manufacturers@chemwhat.com |
Other Suppliers | |
| Watson International Limited | Visit Watson Official Website |
Contact Us for Other Help | |
| Contact us for other information or services | Click here to contact ChemWhat |

