| Official Full Name |
Recombinant Human Fibroblast Growth Factor 19 (rHuFGF-19) |
| Squence |
 |
| Amino Acid Sequence |
MRPLAFSDAG PHVHYGWGDP IRLRHLYTSG PHGLSSCFLR IRADGVVDCA RGQSAHSLLE IKAVALRTVA IKGVHSVRYL CMGADGKMQG LLQYSEEDCA FEEEIRPDGY NVYRSEKHRL PVSLSSAKQR QLYKNRGFLP LSHFLPMLPM VPEEPEDLRG HLESDMFSSP LETDSMDPFG LVTGLEAVRS PSFEK |
| Synonyms |
|
| Accession Number |
O95750 |
| GeneID |
9965 |
| Summary |
|
| Source |
|
| Molecular Weight |
Approximately 21.8 kDa, a single non-glycosylated polypeptide chain containing 195 amino acids. |
| Biological Activity |
Assay #1: Fully biologically active when compared to standard. The biological activity is measured by its binding ability in a functional ELISA. Immobilized rHuFGF R4 at 5 ug/ml can bind rHuFGF-19 with a linear range of 3-200 ng/ml.Assay #2: Fully biologically active when compared to standard. The ED50 as determined by a cell proliferation assay using murine Balb/c 3T3 cells is less than 150 ng/ml, corresponding to a specific activity of > 6.7 × 103 IU/mg. |
| Appearance |
Sterile Filtered White lyophilized (freeze-dried) powder. |
| Formulation |
Lyophilized from a 0.2 um filtered concentrated solution in PBS, pH 7.4. |
| Endotoxin |
Less than 1 EU/ug of rHuFGF-19 as determined by LAL method. |
| Reconstitution |
We recommend that this vial be briefly centrifuged prior to opening to bring the contents to the bottom. Reconstitute in sterile distilled water or aqueous buffer containing 0.1 % BSA to a concentration of 0.1-1.0 mg/mL. Stock solutions should be apportioned into working aliquots and stored at ≤ -20 °C. Further dilutions should be made in appropriate buffered solutions. |
| Stability and Storage |
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.- 12 months from date of receipt, -20 to -70 °C as supplied.- 1 month, 2 to 8 °C under sterile conditions after reconstitution.- 3 months, -20 to -70 °C under sterile conditions after reconstitution. |
| References |
1. Ladher RK, Anakwe KU, Gurney AL, et al. 2000. Science. 290:1965-7.2. Tomlinson E, Fu L, John L, et al. 2002. Endocrinology. 143:1741-7.3. Smallwood PM, Munoz-Sanjuan I, Tong P, et al. 1996. Proc Natl Acad Sci U S A. 93:9850-7.4. Fu L, John LM, Adams SH, et al. 2004. Endocrinology. 145:2594-603.5. Kharitonenkov A, Shiyanova TL, Koester A, et al. 2005. J Clin Invest. 115:1627-35.6. Kurosu H, Kuro OM. 2009. Mol Cell Endocrinol. 299:72-8.7. Lin BC, Wang M, Blackmore C, et al. 2007. J Biol Chem. 282:27277-84.8. Kharitonenkov A, Dunbar JD, Bina HA, et al. 2008. J Cell Physiol. 215:1-7. |
| SDS-PAGE |
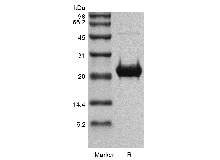 |
| Safety Data Sheet (SDS) Download |
Click to download |
| Technical Data Sheet (TDS) Download |
Click to download |



