| Official Full Name |
Recombinant Human Interferon-gamma GMP (rHuIFN-gamma) |
| Squence |
 |
| Amino Acid Sequence |
MQDPYVKEAE NLKKYFNAGH SDVADNGTLF LGILKNWKEE SDRKIMQSQI VSFYFKLFKN FKDDQSIQKS VETIKEDMNV KFFNSNKKKR DDFEKLTNYS VTDLNVQRKA IHELIQVMAE LSPAAKTGKR KRSQMLFRGR RASQ |
| Synonyms |
Immune Interferon, type II interferon, T cell interferon, MAF |
| Accession Number |
P01579 |
| GeneID |
3458 |
| Summary |
Interferon-gamma (IFN-gamma), also known as Type II interferon or immune interferon, is a cytokine produced primarily by T-lymphocytes and natural killer cells. The protein shares no significant homology with IFN-β or the various IFN-alpha family proteins. Mature IFN-gamma exists as noncovalently-linked homodimers. Human IFN-gamma is highly species specific and is biologically active only in human and primate cells. IFN-gamma was originally characterized based on its antiviral activities. The protein also exerts antiproliferative, immunoregulatory and proinflammatory activities and is thus important in host defense mechanisms. IFN-gamma induces the production of cytokines, upregulates the expression of class I and II MHC antigens, Fc receptor and leukocyte adhesion molecules. It modulates macrophage effector functions, influences isotype switching and potentiates the secretion of immunoglobulins by B cells. IFN-gamma also augments TH1 cell expansion and may be required for TH1 cell differentiation. |
| Source |
Escherichia coli. |
| Molecular Weight |
Approximately 16.9 kDa, a single non-glycosylated polypeptide chain containing 144 amino acids. |
| Biological Activity |
Fully biologically active when compared to standard. The ED50 as measured in anti-viral assays using human HeLa cells infected with encephalomyocarditis (EMC) virus is 0.15-0.80 ng/ml. |
| Appearance |
Sterile filtered white lyophilized (freeze-dried) powder. |
| Formulation |
Lyophilized from a 0.2 um filtered concentrated solution in PBS, pH 5.0, with 3 % Trehalose. |
| Endotoxin |
Less than 0.01 EU/ug of rHuIFN-gamma GMP as determined by LAL method. |
| Reconstitution |
We recommend that this vial be briefly centrifuged prior to opening to bring the contents to the bottom. Reconstitute in sterile distilled water or aqueous buffer containing 0.1 % BSA to a concentration of 0.1-1.0 mg/mL. Stock solutions should be apportioned into working aliquots and stored at ≤ -20 °C. Further dilutions should be made in appropriate buffered solutions. |
| Stability and Storage |
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.- A minimum of 12 months when stored at ≤ -20 °C as supplied. Refer to lot specific COA for the Use by Date.- 1 month, 2 to 8 °C under sterile conditions after reconstitution.- 3 months, -20 to -70 °C under sterile conditions after reconstitution. |
| References |
1. Pennino D, Bhavsar PK, Effner R, et al. 2012. J Allergy Clin Immunol, 2. Hibi M, Hachimura S, Ise W, et al. 2003. Cytotechnology, 43: 49-55.3. Wang H, Ruan Z, Wang Y, et al. 2008. Mol Immunol, 45: 1548-56.4. Kopinski P, Przybylski G, Jarzemska A, et al. 2007. Pol Merkur Lekarski, 23: 15-21. |
| SDS-PAGE |
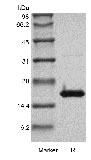 |
| Safety Data Sheet (SDS) Download |
Click to download |
| Technical Data Sheet (TDS) Download |
Click to download |



