Choline glycerophosphate CAS#: 28319-77-9; ChemWhat Code: 285551
Identification
| Product Name | Choline glycerophosphate |
| IUPAC Name | [(2R)-2,3-dihydroxypropyl] 2-(trimethylazaniumyl)ethyl phosphate |
| Molecular Structure | 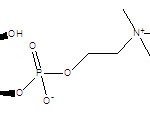 |
| CAS Registry Number | 28319-77-9 |
| EINECS Number | 248-962-2 |
| MDL Number | MFCD00063544 |
| Beilstein Registry Number | No data available |
| Synonyms | L-α-Glycerylphosphorylcholine (2R)-2,3-Dihydroxypropyl 2-(trimethylammonio)ethyl phosphate [ACD/IUPAC Name] (2R)-2,3-Dihydroxypropyl-2-(trimethylammonio)ethylphosphat [German] [ACD/IUPAC Name] (R)-2-[[(2,3-Dihydroxypropoxy)hydroxyphosphinyl]oxy]-N,N,N-trimethylethanaminium hydroxide inner salt CAS# 28319-77-9 CAS No: 28319-77-9 |
| Molecular Formula | C8H20NO6P |
| Molecular Weight | 257.22 |
| InChI | InChI=1S/C8H20NO6P/c1-9(2,3)4-5-14-16(12,13)15-7-8(11)6-10/h8,10-11H,4-7H2,1-3H3/t8-/m1/s1 |
| InChI Key | SUHOQUVVVLNYQR-MRVPVSSYSA-N |
| Canonical SMILES | CN+(C)CCOP(=O)([O-])OCC@@HO |
| Patent Information |
| No data available |
Physical Data
| Appearance | White powder |
| Flash Point | 11°C |
| Melting Point, °C | Solvent (Melting Point) | Comment (Melting Point) |
| 145 – 147 | ethanol, isopropyl alcohol | |
| 142.5 – 143 | ethanol | kryst. bei -15grad. |
| Density, g·cm-3 | Type (Density) |
| 1.32 | crystallographic |
Spectra
| Description (NMR Spectroscopy) | Nucleus (NMR Spectroscopy) | Solvents (NMR Spectroscopy) | Temperature (NMR Spectroscopy), °C | Frequency (NMR Spectroscopy), MHz |
| Spectrum | 1H | 5.84 | ||
| Chemical shifts, Spectrum | 1H | 25 | 700 | |
| Chemical shifts | 1H | dimethylsulfoxide-d6 | ||
| Chemical shifts | 31P | water-d2 | 161.9 | |
| Chemical shifts | 1H | water-d2 | 24.84 | 400.1 |
| Linewidth of NMR absorption | 31P | chloroform-d1 | 26.84 | 162 |
| Chemical shifts | 1H | H2O, NaCl | 37 | 400 |
| Spectrum | 1H | 37 | 400 | |
| Chemical shifts | 1H | D2O, various solvent(s) | ||
| Chemical shifts | 13C | CDCl3, tetradeuteriomethanol | 36 – 38 | |
| Spin-spin coupling constants | CDCl3, tetradeuteriomethanol | 36 – 38 | ||
| Chemical shifts | 14N | D2O |
| Description (IR Spectroscopy) | Solvent (IR Spectroscopy) | Comment (IR Spectroscopy) |
| Spectrum | KBr | 5000 – 667 cm**(-1) |
| Spectrum | nujol | 5000 – 667 cm**(-1) |
| Description (Mass Spectrometry) |
| liquid chromatography mass spectrometry (LCMS), time-of-flight mass spectra (TOFMS), electrospray ionisation (ESI), spectrum |
| time-of-flight mass spectra (TOFMS), electrospray ionisation (ESI), spectrum |
| IT (ion trap), liquid chromatography mass spectrometry (LCMS), electrospray ionisation (ESI), spectrum |
| time-of-flight mass spectra (TOFMS), electrospray ionisation (ESI), spectrum |
| liquid chromatography mass spectrometry (LCMS), high resolution mass spectrometry (HRMS), time-of-flight mass spectra (TOFMS), tandem mass spectrometry, electrospray ionisation (ESI), spectrum |
| liquid chromatography mass spectrometry (LCMS), spectrum |
| electrospray ionisation (ESI), spectrum |
Route of Synthesis (ROS)
| Conditions | Yield |
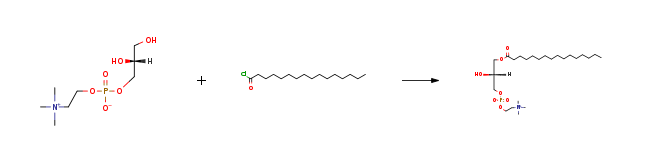
| Stage #1: L-glycero-3-phosphorylcholine With di(n-butyl)tin oxide In isopropyl alcohol for 1h; Heating; Stage #2: n-hexadecanoyl chloride With TEA In isopropyl alcohol at 25℃; for 0.25h; Further stages.; | 97% |
| Stage #1: L-glycero-3-phosphorylcholine With di(n-butyl)tin oxide In isopropyl alcohol for 1h; Reflux; Stage #2: n-hexadecanoyl chloride With triethylamine at 20℃; for 1h; | 90% |
| Stage #1: L-glycero-3-phosphorylcholine With di(n-butyl)tin oxide In isopropyl alcohol for 1h; Heating; Stage #2: n-hexadecanoyl chloride With TEA In isopropyl alcohol at 25℃; for 0.25h; Further stages.; | 80% |
| Stage #1: L-glycero-3-phosphorylcholine With di(n-butyl)tin oxide In isopropyl alcohol for 1h; Reflux; Stage #2: n-hexadecanoyl chloride With triethylamine In isopropyl alcohol at 25℃; for 0.25h; | 80% |
| Stage #1: L-glycero-3-phosphorylcholine With di(n-butyl)tin oxide In isopropyl alcohol at 100℃; for 1h; Stage #2: n-hexadecanoyl chloride With triethylamine In isopropyl alcohol at 20℃; Experimental Procedure 1-O-Hexadecanoyl-sn-glycero-3-phosphocholine (7) General procedure: This compound was prepared as in the literature with some modifications.4 NMR spectra of this compound and of regioisomers can be found elsewhere.5 Migration in compound 7 has been reported when silica gel chromatography was used for purification,6 and sufficient isomeric purity was concluded from 1H- and 13C-NMR in our synthetic protocol. A 100 mL one-neck round-bottom flask equipped with a reflux condenser, was consecutively charged with 2.00 g (8.3 mmol, 1 eq) GPC (6), 2.20 g (8.83 mmol, 1.1 eq) dibutyltin oxide and 80 mL iPrOH. The flask was placed in a pre-heated oil-bath (~100 °C) and it was stirred for 1 h at this temperature. It was cooled to rt, after which 1.3 mL (9.38 mmol, 1.1 eq) Et3N and 2.8 mL (9.2 mmol, 1.1 eq) palmitoyl chloride were added and it was stirred at rt overnight. Subsequently, the mixture was transferred into a separation funnel containing 80 mL H2O and the aqueous layer was washed with pentane (3×80 mL). The aqueous layer was then concentrated in vacuo and the crude product was purified via flash column chromatography (19 g SiO2 gel, 11.8×2.3 cm, 15 mL frac., frac. 6-27 pooled). Yield= 1.28 g (2.58 mmol, 31 %), colorless solid C24H50NO7P [495.63 g·mol-1] Rf= 0.25 (CH2Cl2/MeOH/H2O= 65/25/4, KMnO4) 1H-NMR (300 MHz, MeOD-d4): δ= 4.36-4.24 (m, 2H), 4.24-3.84 (m, 5H), 3.72-3.62 (m, 2H), 3.22 (s, 9H), 2.36 (t, J= 7.5 Hz, 2H), 1.71-1.53 (m, 2H), 1.43-1.19 (m, 24H), 0.90 (t, J= 6.7 Hz, 3H). 13C-NMR (75 MHz, MeOD-d4): δ= 175.3, 69.7, 67.9, 67.5, 66.2, 60.5, 54.7, 34.9, 33.1, 31.0-30.0 (m), 26.0, 23.7, 14.5. Missing signals due to overlap. | 31% |
Safety and Hazards
| GHS Hazard Statements | Not Classified For more detailed information, please visit ECHA C&L website |
| Source: European Chemicals Agency (ECHA) License Note: Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: “Source: European Chemicals Agency, http://echa.europa.eu/”. Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. License URL: https://echa.europa.eu/web/guest/legal-notice Record Name: (1-Cyano-2-ethoxy-2-oxoethylidenaminooxy)dimethylamino-morpholino-carbenium hexafluorophosphate URL: https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/213446 Description: The information provided here is aggregated from the “Notified classification and labelling” from ECHA’s C&L Inventory. Read more: https://echa.europa.eu/information-on-chemicals/cl-inventory-database |
Other Data
| Transportation | Under the room temperature and away from light |
| HS Code | 296200 |
| Storage | Under the room temperature and away from light |
| Shelf Life | 1 year |
| Market Price | USD |
| Druglikeness | |
| Lipinski rules component | |
| Molecular Weight | 257.224 |
| logP | -2.928 |
| HBA | 6 |
| HBD | 2 |
| Matching Lipinski Rules | 4 |
| Veber rules component | |
| Polar Surface Area (PSA) | 108.86 |
| Rotatable Bond (RotB) | 8 |
| Matching Veber Rules | 2 |
| Bioactivity |
| In vitro: Efficacy |
| Quantitative Results |
| pX | Parameter | Value (qual) | Value (quant) | Unit | Target | Dose | Effect |
| 6 | Protection rate | Active | 8.05 | 1 – 10 μM | cardioprotective agent | ||
| 4.1 | Protection rate | Active | 80 μM | cardioprotective agent | |||
| 4 | Protection rate | Active | 100 μM | cardioprotective agent | |||
| 1 | Percentage change(ROS level) | Not active | 1 – 100 μM | cardioprotective agent | |||
| 1 | Percentage change(superoxide anion produced) | Not active | 1 – 100 μM | cardioprotective agent | |||
| percentage(Relative uptake) | = | 73.2 | % | Solute carrier family 22 member 1 [human]:Wild | = 1 mM | ||
| Activity(relative activity) | < | 1 | % | Galactose-3-O-Sulfotransferase [pig]:Wild |
| Quantitative Results | ||
| 1 of 10 | Biological material | macrophage |
| Assay Description | Effect : metabolic Bioassay : LPS: lipopolysaccharide in vitro; effect on NO production assayed; cells from mice fed normal diet for 2 weeks; incub. with title comp. prior to stimulation with Escherichia coli LPS for 24 h; NO production assessed by formation of nitrite measurements | |
| 2 of 10 | Effect | vasoconstrictor agent |
| Assay Description | Target : arteries from Wistar rat Bioassay : isometric tension (IT) stimulated with 1 μM ; IT measured in physiological salt solution (O2:CO2=95:5; pH 7.45-7.50); title comp. effect on stimulated IT studied rings from mesenteric bed of 12-16-w-old male rats mounted in myograph without/with endothelium (-/+E); α-toxin permeabilized by reported method ; title comp. added to bath |
| Toxicity/Safety Pharmacology |
| Quantitative Results |
| pX | Parameter | Value (qual) | Value (quant) | Unit | Target | Effect |
| 6 | increase rate(superoxide anion produced) | Active | Cytotoxic | |||
| 6 | increase rate(ROS level) | Active | Cytotoxic | |||
| 4.67 | IC50 | 21.2 | μM | Phosphoserine phosphatase (gene PSPH) [Caenorhabditis elegans]:Wild | Neurotoxic | |
| 1 | inhibition rate | Not active | ||||
| 1 | increase rate(ROS level) | Active | Cytotoxic | |||
| 1 | Decrease rate | Not active | ||||
| 1 | Percentage change(of cell viability) | Not active | Cytotoxic | |||
| LD50 | = | 650 | mg/kg | |||
| LD50(Lethal dose) | 13000 | mg/kg | ||||
| increase rate(Oxygen consumption rate) | High | Cytotoxic |
| Use Pattern |
| Choline glycerophosphate CAS#: 28319-77-9 is used as Pharmaceuticals |
| Choline glycerophosphate CAS#: 28319-77-9 is treating cognitive dysfunction associated with cerebrovascular diseases |
| Hamartomatous diseases |
| Choline glycerophosphate CAS#: 28319-77-9 Tuberous sclerosis |
| Pulmonary harnartoma |
| von Meyenburg complex |
| Birt-Hogg-Dube syndrome |
| Multiple hemartoma syndrome |
| antioxidant suitable for use in foods, feeds or cosmetics |
| improvement of environmental stress tolerance |
Buy Reagent | |
| No reagent supplier? | Send quick inquiry to ChemWhat |
| Want to be listed here as a reagent supplier? (Paid service) | Click here to contact ChemWhat |
Approved Manufacturers | |
| Caming Pharmaceutical Ltd | http://www.caming.com/ |
| Want to be listed as an approved manufacturer (Requires approvement)? | Please download and fill out this form and send back to approved-manufacturers@chemwhat.com |
Other Suppliers | |
| Watson International Limited | Visit Watson Official Website |
Contact Us for Other Help | |
| Contact us for other information or services | Click here to contact ChemWhat |