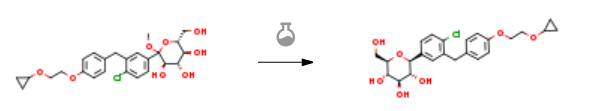
With triethylsilane; boron trifluoride diethyl etherate In dichloromethane; acetonitrile at -25 – -20℃; for 4h; Temperature; Concentration; Inert atmosphere; Large scale;
Experimental Procedure
5 Example 5 Preparation of ((2S,3R,4R,5S,6R)-2-(4-Chloro-3-(4-(2-Cyclopropoxyethoxy)Benzyl)Phenyl)-6-(Hydroxymethyl)Tetrahydro-2H-Pyran-3,4,5-triol
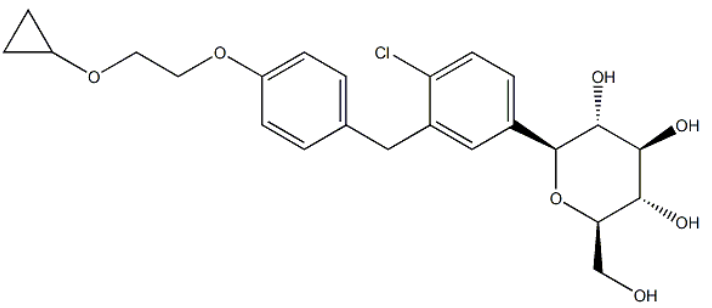
Example 5 Preparation of ((2S,3R,4R,5S,6R)-2-(4-Chloro-3-(4-(2-Cyclopropoxyethoxy)Benzyl)Phenyl)-6-(Hydroxymethyl)Tetrahydro-2H-Pyran-3,4,5-triol [0236] This example describes preparation of (2S,3R,4R,5S,6R)-2-(4-chloro-3-(4-(2-cyclopropoxyethoxy)benzyl)phenyl)-6-(hydroxymethyl)tetrahydro-2H-pyran-3,4,5-triol by reduction of the anomeric OMe and/or OH. (3R,4S,5S,6R)-2-(4-Chloro-3-(4-(2-Cyclopropoxyethoxy)Benzyl)Phenyl)-6-(Hydroxymethyl)-2-Methoxytetrahydro-2H-Pyran-3,4,5-Triol Solution [0237] A 30 L glass reactor equipped with a thermometer was charged with crude (3R,4S,5S,6R)-2-(4-chloro-3-(4-(2-cyclopropoxyethoxy)benzyl)phenyl)-6-(hydroxymethyl)-2-methoxytetrahydro-2H-pyran-3,4,5-triol (2.7 kg), DCM (5.4 kg) and acetonitrile (3.2 kg), and the mixture was magnetically stirred until all the solids dissolved under nitrogen sparging. [0238] Triethylsilane Solution: [0239] BF3.Et2O (2.34 kg) was added to a cold (-21 to -15° C.) solution of triethysilane (2.55 kg) dichloromethane (5.4 kg) and acetonitrile (3.2 kg) under nitrogen. [0240] The (3R,4S,5S,6R)-2-(4-chloro-3-(4-(2-cyclopropoxyethoxy)benzyl)phenyl)-6-(hydroxymethyl)-2-methoxytetrahydro-2H-pyran-3,4,5-triol solution was added to the cold triethylsilane solution at such a rate to maintain the temperature between -20 and -25° C. (3 h). [0241] The reaction mixture was stirred for another 4 h at -22 to -25° C. and then quenched by addition of an aqueous solution of sodium bicarbonate (7.4% w/w, 18.3 kg) while keeping the internal temperature below -10° C. Solid sodium bicarbonate (1.35 kg) was added to adjust the pH to 7.5. The solvents were removed under reduced pressure (temperature below 40° C.). The residue was partitioned between ethyl acetate (18 kg) and water (9.2 kg). The layers were separated and the aqueous layer was extracted with ethyl acetate (2×9 kg). The combined organic layers were washed with brine (2×9 kg) and the solvents were removed under reduced pressure at the temperature below 40° C. until the condensation almost stop Anhydrous ethanol (9 kg) was added and concentrated to give the crude product of the title compound (2.5 kg, 90% yield, 90.8% HPLC purity, HPLC-0001) as foamy solid. | 90% |