Ethyl (R)-3-hydroxybutyrate CAS#: 24915-95-5; ChemWhat Code: 247203
Identification
| Patent Information | ||
| Patent ID | Title | Publication Date |
| US2022/110905 | Efficient anti-bacterial hydroxy acid ester oligomer | 2022 |
| US2021/94900 | SYNTHESIS OF 3-HYDROXYBUTYRYL 3-HYDROXYBUTYRATE AND RELATED COMPOUNDS | 2021 |
| KR2019/26747 | Beta – hydroxy butyrate and butanediol compositions and methods of using method-chain fatty acid ester and methods of using | 2019 |
| US2015/210665 | ORGANOCATALYTIC PROCESS FOR ASYMMETRIC SYNTHESIS OF DECANOLIDES | 2015 |
Physical Data
| Appearance | White or off-white powder |
| Boiling Point, °C | Pressure (Boiling Point), Torr |
| 66 | 8 |
| 85 – 89 | 2 |
| 87.1 – 87.7 | 26 |
| 81 – 83 | 20 |
| 84 – 85 | 20 |
| 86 – 86.5 | 22 |
| 74 | 12 |
| Description (Association (MCS)) | Temperature (Association (MCS)), °C | Partner (Association (MCS)) |
| Further physical properties of the adsorbed molecule. | 32.85 | Ni3[C6H3(COOH)3]2(3-picoline)6(1,2-propanediol)12(H2O)11 |
Spectra
| Description (NMR Spectroscopy) | Nucleus (NMR Spectroscopy) | Solvents (NMR Spectroscopy) | Frequency (NMR Spectroscopy), MHz |
| Chemical shifts, Spectrum | 1H | chloroform-d1 | |
| Chemical shifts | 1H | chloroform-d1 | |
| Chemical shifts, Spectrum | 13C | chloroform-d1 | 101 |
| Chemical shifts, Spectrum | 1H | dimethylsulfoxide-d6 | 400 |
| Chemical shifts | 1H | chloroform-d1 | 400 |
| Chemical shifts | 13C | chloroform-d1 | 100 |
| Chemical shifts, Spectrum | 1H | chloroform-d1 | 400 |
| Description (IR Spectroscopy) | Solvent (IR Spectroscopy) |
| Bands | |
| Bands | neat liquid |
| ATR (attenuated total reflectance), Bands | |
| Bands | chloroform |
| Bands | chloroform |
| Bands | chloroform |
Route of Synthesis (ROS)
| Conditions | Yield |
| With lithium aluminium tetrahydride In diethyl ether for 1h; Ambient temperature; | 86% |
| With lithium aluminium tetrahydride In diethyl ether at 0℃; for 0.5h; | 85.6% |
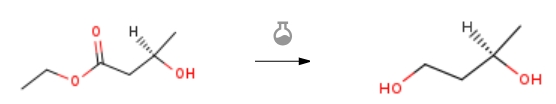
| Stage #1: Ethyl (R)-3-hydroxybutanoate With lithium aluminium tetrahydride In diethyl ether at 0 – 20℃; for 3h; Inert atmosphere; Stage #2: With water In diethyl ether Experimental Procedure 5.2. General procedure for the synthesis of 1,3-diols from β-hydroxyesters General procedure: This method was adapted from the literature.14 To a suspension of LiAlH4 (7.6 mmol, 1 equiv) in dry diethyl ether (6 mL) was added a solution of β-hydroxyester (7.6 mmol, 1 equiv) in dry diethyl ether (5 mL) at 0 °C. The mixture was stirred for 3 h at room temperature and then quenched with distilled water (1 mL). After 10 mn, a 30% NaOH solution (1.5 mL) was added followed by 1 mL of distilled water. The reaction mixture was passed through a Celite pad. The filtrate was dried over MgSO4, concentrated under vacuum and purified by column chromatography if necessary (silica gel, cyclohexane/ethyl acetate 20/80) to afford the desired diol. | 81% |
Safety and Hazards
| Pictogram(s) |  |
| Signal | Warning |
| GHS Hazard Statements | H227 (50%): Combustible liquid [Warning Flammable liquids] H315 (100%): Causes skin irritation [Warning Skin corrosion/irritation] H319 (100%): Causes serious eye irritation [Warning Serious eye damage/eye irritation] H335 (100%): May cause respiratory irritation [Warning Specific target organ toxicity, single exposure; Respiratory tract irritation] H413 (50%): May cause long lasting harmful effects to aquatic life [Hazardous to the aquatic environment, long-term hazard] |
| Precautionary Statement Codes | P210, P261, P264, P264+P265, P271, P273, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P370+P378, P403, P403+P233, P405, and P501 (The corresponding statement to each P-code can be found at the GHS Classification page.) |
Other Data
| Transportation | Under room temperature away from light |
| HS Code | |
| Storage | Under room temperature away from light |
| Shelf Life | 1 year |
| Market Price |
| Druglikeness | |
| Lipinski rules component | |
| Molecular Weight | 132.159 |
| logP | 0.245 |
| HBA | 3 |
| HBD | 1 |
| Matching Lipinski Rules | 4 |
| Veber rules component | |
| Polar Surface Area (PSA) | 46.53 |
| Rotatable Bond (RotB) | 4 |
| Matching Veber Rules | 2 |
| Use Pattern |
| EthylR-3-hydroxybutyrate CAS 24915-95-5 is a versatile multidentate ligand commonly used as a metal chelating agent. Its strong chelation properties make it ideal for various applications involving metal ion analysis and testing. In spectrophotometric methods, it is used to determine the concentrations of metals such as iron, copper, cobalt, and chromium. For iron and manganese, it plays a crucial role in iodometric titrations, providing reliable measurements. Furthermore, this compound is applied in the rapid determination of calcium in bones and teeth, aiding in biological and medical studies. Its utility extends to complexometric titrations, where it is used for the precise measurement of thorium and sulfates. Overall, trans-1,2-Cyclohexanediaminetetraacetic acid monohydrate is an essential reagent in various fields, including analytical chemistry, biology, and environmental science, due to its ability to form stable complexes with a wide range of metal ions, ensuring accuracy and precision in quantitative analysis. |
Buy Reagent | |
| No reagent supplier? | Send quick inquiry to ChemWhat |
| Want to be listed here as a reagent supplier? (Paid service) | Click here to contact ChemWhat |
Approved Manufacturers | |
| Want to be listed as an approved manufacturer (Requires approvement)? | Please download and fill out this form and send back to approved-manufacturers@chemwhat.com |
Other Suppliers | |
| Watson International Limited | Visit Watson Official Website |
Contact Us for Other Help | |
| Contact us for other information or services | Click here to contact ChemWhat |