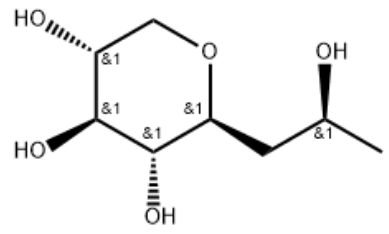
(S)-Pro-xylane CAS#: 868156-46-1; ChemWhat Code: 1417895
Identification
| Patent Information | ||
| Patent ID | Title | Publication Date |
| US2005/250708 | Novel C-glycosides, uses thereof | 2005 |
Physical Data
| Appearance | Solid |
| Melting Point, °C |
| 120 – 122 |
Spectra
| Description (NMR Spectroscopy) | Nucleus (NMR Spectroscopy) | Solvents (NMR Spectroscopy) |
| Chemical shifts, Spectrum | 1H | water-d2 |
| Chemical shifts, Spectrum | 13C | water-d2 |
| Chemical shifts | 1H | |
| Chemical shifts | 13C | |
| Chemical shifts | 1H | CD3OD |
Route of Synthesis (ROS)
| Conditions | Yield |
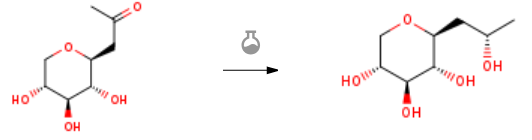
| With sodium tris(acetoxy)borohydride In isopropyl alcohol at 20℃; Product distribution / selectivity; Experimental Procedure 2 Production of C-β-D-xylopyranoside-2-(S)-hydroxypropane EXAMPLE 2 Production of C-β-D-xylopyranoside-2-(S)-hydroxypropane The expected compound was obtained according the process described in Example 1, replacing the 2 ml of acetic acid and the sodium borohydride with two times 1.2 equivalents of sodium triacetoxyborohydride (NaBH(OAc)3). The product was obtained with a quantitative yield, and selectively in (β, S) form. The physico chemical characteristics are identical in all respects to those obtained in Example 1. | 100% |
| With sodium tetrahydroborate; acetic acid In isopropyl alcohol at 20℃; for 1.5h; Product distribution / selectivity; Experimental Procedure 1 Production of C-β-D-xylopyranoside-2-(S)-hydroxypropane EXAMPLE 1 Production of C-β-D-xylopyranoside-2-(S)-hydroxypropane 2 ml of acetic acid, followed rapidly by 120 mg (1.2 eq.) of sodium borohydride as granules were added to a solution of 500 mg of C-β-D-xylopyranoside-2-propanone (described in Example 1 of application WO-02/051828) in 9 ml of isopropanol. The medium was left at ambient temperature for 30 minutes with stirring. 120 mg (1.2 eq.) of sodium borohydride as granules were then added. The reaction medium was left for 1 hour at ambient temperature with stirring. 10 ml of acetone were then added and, after stirring for 30 minutes at ambient temperature, the reaction medium was concentrated under vacuum. The residue obtained was purified by silica gel chromatography so as to selectively produce the expected compound C-β-D-xylopyranoside-2-(S)-hydroxypropane with a 95% yield. Physico chemical characteristics of the compound: Melting point: 120-122° C. Optical rotation: -37° (at 20° C. in methanol, and at a concentration [C]=1 g/100 ml) 1H NMR: 1.03 (t, 3H); 1.46 (m, 1H); 1.71 (m, 1H); 2.85 (m, 1H); 2.94 (m, 1H); 2.99 (m, 2H); 3.24 (m, 1H); 3.67 (m, 1H); 3.77 (m, 1H) Structure confirmed by X-ray diffraction. | 95% |
Safety and Hazards
| Pictogram(s) | |
| Signal | |
| GHS Hazard Statements | Not Classified |
| Precautionary Statement Codes |
Other Data
| Druglikeness | |
| Lipinski rules component | |
| Molecular Weight | 192.212 |
| logP | -1.202 |
| HBA | 5 |
| HBD | 4 |
| Matching Lipinski Rules | 4 |
| Veber rules component | |
| Polar Surface Area (PSA) | 90.15 |
| Rotatable Bond (RotB) | 2 |
| Matching Veber Rules | 2 |
| Use Pattern |
| Anti-aging, promotes skin cell renewal, helps repair and rebuild the skin’s collagen and glycosaminoglycans, thereby reducing wrinkles and fine lines. Moisturizing and nourishing, promotes the synthesis of collagen, repairs damaged skin; enhances overall skin health. |
Related Chemicals
Buy Reagent | |
| No reagent supplier? | Send quick inquiry to ChemWhat |
| Want to be listed here as a reagent supplier? (Paid service) | Click here to contact ChemWhat |
Approved Manufacturers | |
| Apnoke Scientific Limited | http://www.apnoke.com/ |
| Want to be listed as an approved manufacturer (Requires approvement)? | Please download and fill out this form and send back to approved-manufacturers@chemwhat.com |
Other Suppliers | |
| Watson International Limited | Visit Watson Official Website |
Contact Us for Other Help | |
| Contact us for other information or services | Click here to contact ChemWhat |