Sulfonium, triphenyl-, salt with 3,3,4,4-tetrafluoro-4-sulfobutyl tricyclo[3.3.1.13,7]decane-1-carboxylate (1:1) CAS#: 1001347-91-6; ChemWhat Code: 1497948
Identification
| Product Name | Sulfonium, triphenyl-, salt with 3,3,4,4-tetrafluoro-4-sulfobutyl tricyclo[3.3.1.13,7]decane-1-carboxylate (1:1) |
| IUPAC Name | 4-(adamantane-1-carbonyloxy)-1,1,2,2-tetrafluorobutane-1-sulfonate;triphenylsulfanium |
| Molecular Structure | 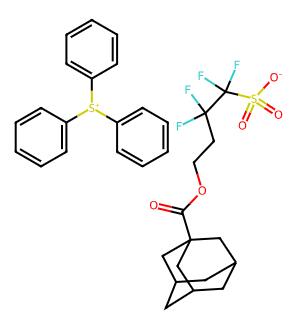 |
| CAS Registry Number | 1001347-91-6 |
| EINECS Number | No data available |
| MDL Number | No data available |
| Beilstein Registry Number | No data available |
| Synonyms | triphenylsulfonium ((adamantane-1-carbonyl)oxy)-tetrafluorobutanesulfonate |
| Molecular Formula | C33H34F4O5S2 |
| Molecular Weight | 650.742 |
| InChI | InChI=1S/C18H15S.C15H20F4O5S/c1-4-10-16(11-5-1)19(17-12-6-2-7-13-17)18-14-8-3-9-15-18;16-14(17,15(18,19)25(21,22)23)1-2-24-12(20)13-6-9-3-10(7-13)5-11(4-9)8-13/h1-15H;9-11H,1-8H2,(H,21,22,23)/q+1;/p-1 |
| InChI Key | InChI=1S/C18H15S.C15H20F4O5S/c1-4-10-16(11-5-1)19(17-12-6-2-7-13-17)18-14-8-3-9-15-18;16-14(17,15(18,19)25(21,22)23)1-2-24-12(20)13-6-9-3-10(7-13)5-11(4-9)8-13/h1-15H;9-11H,1-8H2,(H,21,22,23)/q+1;/p-1 |
| Canonical SMILES | O=C(OCCC(F)(F)C(F)(F)S(=O)(=O)[O-])C12CC3CC(CC(C3)C1)C2.c1ccc(S+c2ccccc2)cc1 |
| Patent Information | ||
| Patent ID | Title | Publication Date |
| CN119264025 | Preparation method of photoacid generator for semiconductor photoresist | 2025 |
| TW2024/23898 | Radiation-sensitive resin composition, pattern formation method, and radiation-sensitive acid-generating agent | 2024 |
Physical Data
| Appearance | White to off-white powder |
| Solubility | No data available |
| Flash Point | No data available |
| Refractive index | No data available |
| Sensitivity | No data available |
Spectra
| Description (NMR Spectroscopy) | Nucleus (NMR Spectroscopy) |
| Chemical shifts, Spectrum | 1H |
Route of Synthesis (ROS)
![Route of Synthesis (ROS) of Sulfonium, triphenyl-, salt with 3,3,4,4-tetrafluoro-4-sulfobutyl tricyclo[3.3.1.13,7]decane-1-carboxylate (11) CAS# 1001347-91-6](https://www.chemwhat.com/wp-content/uploads/2026/04/Route-of-Synthesis-ROS-of-Sulfonium-triphenyl-salt-with-3344-tetrafluoro-4-sulfobutyl-tricyclo3.3.1.137decane-1-carboxylate-11-CAS-1001347-91-6.jpg)
| Conditions | Yield |
| With trifluoroacetic acid; trifluoroacetic anhydride at -20 – 40℃; for 12h; Temperature; Inert atmosphere; Experimental Procedure 1.2-1.3; 6.2-6.3; 7.2-7.3; 8.2-8.3; 9.2-9.3; 10.2-10.3; 11.2-11.2; 12.2-12.3; 13.2-13.3; 14.2-14.3; 15.2-15.3; 16.2-16.3; 17.2-17.3; 2.2-2.3; 3.2-3.3; 4.2-4.3; 5.2-5.3 S2. The reactor was purged with nitrogen, 148.0 g of the intermediate product 1,1,2,2-tetrafluoro-4-hydroxybutane-1-sulfonic acid triphenylsulfonium salt, 60.0 g of adamantane carboxylic acid and 624.1 g of trifluoroacetic acid were added and stirred to dissolve, the reaction temperature was lowered to -20 °C, 127.2 g of trifluoroacetic anhydride was added dropwise, and after the addition was completed, the temperature was raised to 40 °C and the reaction was carried out for 12 h. After the reaction was completed, the reaction was distilled under reduced pressure to obtain a concentrated reaction solution; S3. In the concentrated reaction solution prepared in S2, 2 kg of methyl tert-ether was added as a precipitating solvent, and recrystallized, stirred for 4 h, and then filtered to obtain 177.8 g of a photoacid generator; | 90.2% |
Safety and Hazards
| GHS Hazard Statements | Not Classified |
Other Data
| Transportation | NONH for all modes of transport |
| Under the room temperature and away from light | |
| HS Code | No data available |
| Storage | Under the room temperature and away from light |
| Shelf Life | 1 year |
| Market Price | USD |
| Druglikeness | |
| Lipinski rules component | |
| Molecular Weight | 650.756 |
| logP | 9.792 |
| HBA | 5 |
| HBD | 2 |
| Matching Lipinski Rules | 2 |
| Veber rules component | |
| Polar Surface Area (PSA) | 51.21 |
| Rotatable Bond (RotB) | 10 |
| Matching Veber Rules | 2 |
| Use Pattern |
| Serves as a photoacid generator (PAG) in chemically amplified photoresists, mainly used for semiconductor lithography processes such as KrF (248 nm) and ArF (193 nm) exposure, to realize high-resolution pattern formation for integrated circuits and advanced chip manufacturing. |
| Applied in electronics imaging materials including LCD panels, printing plates, photocurable coatings and inks, providing high sensitivity and thermal stability. |
| Used as a photochemical reagent in organic synthesis and laboratory R&D, for light-triggered acid-catalyzed reactions in fine chemical preparation. |
Buy Reagent | |
| No reagent supplier? | Send quick inquiry to ChemWhat |
| Want to be listed here as a reagent supplier? (Paid service) | Click here to contact ChemWhat |
Approved Manufacturers | |
| WatsonChem Advanced Chemical Materials | https://www.watsonchem.com/ |
| Want to be listed as an approved manufacturer (Requires approvement)? | Please download and fill out this form and send back to approved-manufacturers@chemwhat.com |
Other Suppliers | |
| Watson International Limited | Visit Watson Official Website |
Contact Us for Other Help | |
| Contact us for other information or services | Click here to contact ChemWhat |
